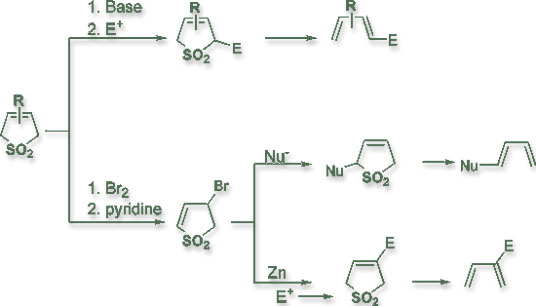
As a research fellow at Academia Sinica, Dr. Chou has devoted to the chemistry of 3-sulfolenes for more than a decade. In 1984, Dr. Chou began the study of deprotonation/substitution reaction of 3-sulfolene and developed the reaction to an ideal methodology of using 3-sulfolene as the precursor for butadienyl-1-anion. The regioselectivity nature of the substitution reaction and the stereospecific nature of the extrusion of sulfur dioxide from 3-sulfolenes rapidly made 3-sulfoelnes building blocks for the construction of interesting organic molecules. When the deprotonation/substitution sequence is accompanied by subsequent intramolecular Diels-Alder reactions, a variety of bicyclic and multicyclic skeleton can be constructed. This strategy has been utilized not only by Dr. Chou but also by other research groups in the world as the key steps to synthesize complex natural products. Further more, Dr. Chou extended the study and made 3-sulfolenes also as equivalents to butadienyl-2-anion, butadienyl-1,4-dianion, butadienyl-1,1-dianion, butadienyl-1-cation, butadienyl-2-cation, and so on. Therefore a wide variety of substituted butadienes can be conveniently prepared from 3-sulfolene. Dr. Chou was invited by the 4ACC to deliver a lecture in Beijing to review his accomplishment in this area.
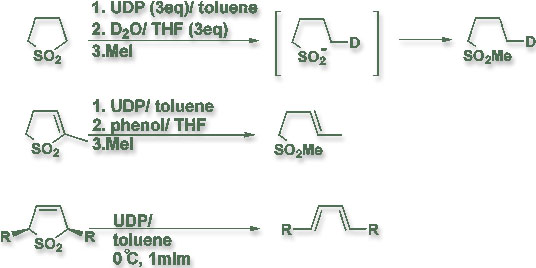
Dr. Chou's research also covers ultrasound chemistry, especially the use of ultrasonically dispersed potassium (UDP) in organic synthesis. UDP was found to be an efficient reducing agent for C-S bond cleavage of sulfones as well as a promoter for cheletropic extrusion of sulfur dioxide from substituted 3-sulfolenes. The regio- and stereoselectivity of the C-S bond cleavage made UDP a reagent for the preparation of useful intermediates for organic synthesis. An especially interesting point in this study is the finding that UDP transfers to sulfone functionality much faster than its reaction with an acidic proton. Dr. Chou also utilized ultrasound to promote the lithium-mediated C-P bond cleavage reactions and developed efficient approaches toward the preparation of symmetric and unsymmetric DIPHOS ligands. Due to his success in this area of research, Dr. Chou was invited as an international advisory editor of the journal "Ultrasound: Sonochemistry".
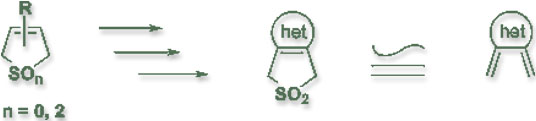
The more recent achievement of Dr. Chou's research is in the area of heteroaromatic o-quinodimethanes. Dr. Chou has developed several general synthetic approaches , each of which can be used to prepare a variety of herteroaromatic-fused 3-sulfolenes, the ideal precursors for the corresponding o-quinodimethanes. Beforehand, the chemistry of heteroaromatic o-quinodimethanes was interested mainly by physical organic chemists, but not so by synthetic organic chemists. Dr. Chou's research results demonstrated that functionalization of the precursors can be easily achieved, so that their synthetic applications toward the construction of complex multicyclic heterocycles are now possible. The review article "Heteroaromatic o-Quinodimethanes" authored by Dr. Chou [Rev. Heteroatom Chem, 8, 65-104 (1993)] is perhaps the most frequently cited reference in this field of research. Dr. Chou is invited by the Organic Chemistry Division of the 7ACC in Hiroshima to present an invited lecture on his recent progress in this area.
Having served as the Director of the Institute of Chemistry, Academia Sinica for nine years (1987-1996) and the President of the Chemical Society in Taipei for two years (1993-1994), Dr. Chou put in great efforts to promote the international activities of the chemistry community in Taiwan. In the last decade, the R.O.C. government gave fairly strong support to basic research and the quality of chemistry research improved significantly. International activities not only further stimulate the domestic chemists to work harder, but also help foreign chemists to notice the development in this area. Among the most successful activities are: